Zinc Sulfate Monohydrate
Zinc Sulfate Monohydrate is a moderately water and acid soluble Zinc source for uses compatible with sulfates. Sulfate compounds are salts or esters of sulfuric acid formed by replacing one or both of the hydrogens with a metal. Most metal sulfate compounds are readily soluble in water for uses such as water treatment. Organometallic forms are soluble in organic solutions and sometimes in both aqueous and organic solutions. Metallic ions can also be dispersed utilizing suspended or coated nanoparticles and deposited utilizing sputtering targets and evaporation materials for uses such as solar cells and fuel cells. Zinc Sulfate Monohydrate is generally immediately available in most volumes. High purity, submicron and nanopowder forms may be considered.
Specification:
| Formula | ZnSO4·H2O |
| Purity: | 98% |
| Zn: | 35.5%min |
| Pb: | 10ppm max |
| Cd: | 10ppm max |
| As: | 5ppm max |
| Insoluble: | 0.05% max |
Application Overview
-Zinc sulfate monohydrate is used in calico printing, wood and skin preservation, galvanizing electrolytes, bleached paper and clear glue.
-Chemical reagents in industry, coagulants in rayon preparation, mordants in dyeing, and zinc sources in animal feed.
-Medically, it is used as an astringent and emetic. Mono zinc sulphate is the precursor of pigment lithopone.
-Monohydrate zinc sulfate is also used to provide zinc in fertilizers, agricultural sprays, galvanizing electrolytes, and as a mordant in dyeing.
Related Elements

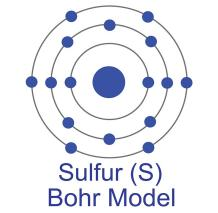
Sulfur (or Sulphur) (atomic symbol: S, atomic number: 16) is a Block P, Group 16, Period 3 element with an atomic radius of 32.066.In its elemental form, sulfur has a light yellow appearance. The sulfur atom has a covalent radius of 105 pm and a Van der Waals radius of 180 pm. In nature, sulfur can be found in hot springs, meteorites, volcanoes, and as galena, gypsum, and epsom salts. Sulfur has been known since ancient times but was not accepted as an element until 1777, when Antoine Lavoisier helped to convince the scientific community that it was an element and not a compound.

Zinc (atomic symbol: Zn, atomic number: 30) is a Block D, Group 12, Period 4 element with an atomic weight of 65.38. The number of electrons in each of zinc's shells is 2, 8, 18, 2, and its electron configuration is [Ar] 3d10 4s2. The zinc atom has a radius of 134 pm and a Van der Waals radius of 210 pm. Zinc was discovered by Indian metallurgists prior to 1000 BC and first recognized as a unique element by Rasaratna Samuccaya in 800. Zinc was first isolated by Andreas Marggraf in 1746. In its elemental form, zinc has a silver-gray appearance. It is brittle at ordinary temperatures but malleable at 100 °C to 150 °C. It is a fair conductor of electricity, and burns in air at high red producing white clouds of the oxide. Zinc is mined from sulfidic ore deposits. It is the 24th most abundant element in the earth's crust and the fourth most common metal in use). The name zinc originates from the German word "zin," meaning tin.

Why Choose Us
Reliable
We have handled chemical.additives for 9 years.And enjoy good reputation in the world market for our good quality and reasonable prices.A partner you can trust.
A wide range of products
We are familiar with the domestinc raw material market and are involved in the business of ferrous sulfate,copper sulfate ammonium sulfate,and all Sulphate salts.
Rich resources
We have two factories which is specialized in zinc sulphate and manganese sulphate.Over 100000tons per year.Ensure sufficient supply for customers.
Strong communication skills and service ethic
As the agent of factory,our team have same expertise as the factory but stronger communication skills to improve the efficiency of negotiations.
Competitive Advantages of Our Company
WIT-STONE cooperates with reputable large manufacturers in the raw material procurement of zinc sulfate monohydrate. After the raw materials are purchased into the factory, the raw materials shall be inspected first, and then the raw materials warehouse shall be coded and stacked for quality tracking in the future. WIT-STONE has purchased the most advanced production equipment and testing equipment of zinc sulfate monohydrate in the world to provide customers with the best quality products and services. Before production, the raw material zinc oxide shall be rinsed; During the production process, multi-effect evaporator and hot-air dryer are used for evaporation and drying, which is environmentally friendly and efficient. After the production of the product is completed, the finished product is inspected by atomic absorption spectrophotometer and polarographic analyzer, and can be delivered only after passing the inspection.
In addition, some customers asked about the causes of zinc sulfate caking, mainly including:
1. The raw materials are not rinsed during production, and the chloride ion content is too high, which is easy to agglomerate;
2. The temperature of the zinc sulfate produced is very high. Many manufacturers fill the zinc sulfate too early due to rush or site reasons, which leads to high temperature in the packaging bag. In addition, there is no ventilation or high temperature during long-distance transportation, which causes zinc sulfate agglomeration.
In order to effectively solve the problem of zinc sulfate agglomeration, Changsha Ruiqi Chemical Products Co., Ltd. will add a rinsing process after purchasing raw materials to remove chloride ions in raw materials; For zinc sulfate monohydrate, a new drying procedure is added to the original process to reduce the surface moisture of zinc sulfate monohydrate and avoid agglomeration during transportation.
Production method of our company:
The company's production process method is that zinc oxide reacts with sulfuric acid solution to form a first stage acid leaching solution and a first stage acid leaching residue, adding hydrogen peroxide and zinc oxide to the first stage acid leaching solution to oxidize and precipitate iron, adding the first stage acid leaching residue to sulfuric acid solution for second stage acid leaching, and then pressing filtration to form a second stage acid leaching solution and a second stage acid leaching residue, adding scrap iron and P204 to the second stage acid leaching solution, and reacting the second stage acid leaching solution with zinc oxide, Conduct iron removal and neutralization, add zinc powder for replacement and purification, and then add the secondary acid leaching solution that has been replaced and purified into the primary acid leaching solution. The zinc sulfate monohydrate crystal is obtained by three-effect evaporation crystallization using hot steam. This production process improves the zinc content in the acid leaching solution and reduces the cadmium content in the acid leaching solution, which not only improves the product quality, but also improves the utilization rate of raw materials and product output rate; At the same time, the three-effect evaporation crystallization of acid leaching solution is adopted to reduce the heat vapor required for evaporation crystallization, thus reducing the heat consumption.
Packaging details:
25kg,50kg,1000kg, 1250kg,Container bag and OEM color bag
Inside double resealable zip bags and outside with aluminium Foil Bags OR bigger size double seal PET bags for 25kgs in bulk then packed in drums for shipping.
Shipment:
Support various modes of transportation, welcome to contact us for consultation.
shipping: will be about 7-15days after receive the payment .
Port:Any Port in China
Storage:
Zinc Sulphate Should be stored in cool and ventilated place ,keep away from fire, heat and sunshine, sealed package. Stay away from oxide.

I’m glad to meet WIT-STONE, who is really an excellent chemical supplier. Cooperation needs to continue, and trust is built little by little. They have a strict quality control system, which I highly appreciate.
After selecting Zinc sulfate monohydrate suppliers for many times, we resolutely chose WIT-STONE. Integrity, enthusiasm and professionalism have captured our trust again and again.


Stating easy process. Great customer service. Process from ordering to delivery was easy. WIT-STONE provided excellent customer service. Delivery was on time and an update email was provided to me during each step of the process. Nicely done.
Q: How can I know whether your performance is better?
A: My friend, the best way to check whether the performance is good or not good is to get some samples to test.
Q:Can l get a lower price if I order large quantity?
A:Yes, prices discount according to the order quantity and payment term.
Q:Can you arrange third party testing of the chemical before purchase of zinc sulfate?
A:Yes We work with reputable international testing agencies such as SCS Bureau Veritas, Intertek CCIC and other agencies that clients worldwide trust to conduct independent testing. We arrange for the agencies to visit the plant. review production. test product, issue reports and seal containers before export.
Q:Do vou arrange for certificate of conformity (COC) and pre-export verification document (pvoc)?
A:Again working with the international agencies that are authorized to conduct COC/PVOC for our country. we will arrange COC /PVOC in compliance with your country's request. Please note that extra COC/PVOC cost do apply.
Q: Will my cargo be insured in transit?
A:Yes, under international terms of CIF. all chemicals are insured with top global insurance agencies.
Q: Do you accept bulk and small orders of zinc sulfate?
A:WIT-STONE is experienced at managing bulk orders for all zinc sulfate. WIT-STONE does engage in small - scale orders in order to help our clients scale to larger orders or have samples for testing. However, our major focus in on orders greater than 1 20ft container.











